Research
Unravelling the Arthus Mystery: Fc-Receptors and the Holy Grail of Inflammation A 32-minute presentation by Dr. Ravetch and one of his previous trainees (now Professor and Chair of Genetics at Friedrich Alexander University of Erlangen-Nürnberg) present “Unravelling the Arthus Mystery: Fc-Receptors and the Holy Grail of Inflammation.
A 32-minute presentation by Dr. Ravetch and one of his previous trainees (now Professor and Chair of Genetics at Friedrich Alexander University of Erlangen-Nürnberg) present “Unravelling the Arthus Mystery: Fc-Receptors and the Holy Grail of Inflammation.
IgG Rc Receptors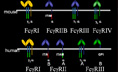 The first cloning of Fc receptors was reported by the Ravetch lab in 1986 and subsequent studies have revealed a multi-gene family of oligomeric receptors for the Fc domain of antibodies. Mice deficient in specific FcRs were generated by that group in 1994 and has lead to a detailed understanding of the role of these receptors in inflammation and modulation of the immune response.
The first cloning of Fc receptors was reported by the Ravetch lab in 1986 and subsequent studies have revealed a multi-gene family of oligomeric receptors for the Fc domain of antibodies. Mice deficient in specific FcRs were generated by that group in 1994 and has lead to a detailed understanding of the role of these receptors in inflammation and modulation of the immune response.
FcRs and TOLERANCE RIIB-/- mice spontaneously develop autoantibodies and autoimmune glomerulonephritis in a strain-dependent fashion. C57Bl/6 mice deficient in this inhibitory receptor develop autoantibodies and autoimmune glomerulonephritis, while the same genetic lesion on the Balb/c backgrond does not result in a spontaneous phenotype.
RIIB-/- mice spontaneously develop autoantibodies and autoimmune glomerulonephritis in a strain-dependent fashion. C57Bl/6 mice deficient in this inhibitory receptor develop autoantibodies and autoimmune glomerulonephritis, while the same genetic lesion on the Balb/c backgrond does not result in a spontaneous phenotype.
Activation FcyR Are Required for the Development of Tissue Pathology in Autoimmune Glomerulonephritis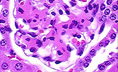 A spontaneous model of lupus, the NZBxNZW f1, develops fatal glomerulonephritis as a consequence of immune complex deposition in the kidney. Deletion of the activation FcR g chain protects this susceptible strain from inflammatory disease, while not affecting immune complex deposition or C3 deposition.
A spontaneous model of lupus, the NZBxNZW f1, develops fatal glomerulonephritis as a consequence of immune complex deposition in the kidney. Deletion of the activation FcR g chain protects this susceptible strain from inflammatory disease, while not affecting immune complex deposition or C3 deposition.
Therapeutic IgG cytotoxicity requires activation FcR The anti-CD20 antibody, rituximab, approved for the treatment of non-Hodgkins lymphoma, requires activation FcRs to mediate its anti-tumor effect. Mice bearing human B cell lymphomas were successfully able to arrest tumor growth in response to rituximab treatment if they expressed activation FcRs.
The anti-CD20 antibody, rituximab, approved for the treatment of non-Hodgkins lymphoma, requires activation FcRs to mediate its anti-tumor effect. Mice bearing human B cell lymphomas were successfully able to arrest tumor growth in response to rituximab treatment if they expressed activation FcRs.
Improving Vaccination Via DC Targeting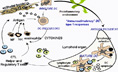 Dendritic cells capture antigens and present them to cognate T cells to initiate the cascade of cellular events ultimately leading to effective immunity. However, antigen capture by DCs alone will not result in T cell activation unless a "maturation" stimulus is provided to the DC to induce co-stimulatory molecules.
Dendritic cells capture antigens and present them to cognate T cells to initiate the cascade of cellular events ultimately leading to effective immunity. However, antigen capture by DCs alone will not result in T cell activation unless a "maturation" stimulus is provided to the DC to induce co-stimulatory molecules.
IVIG Protects from K/BxN Arthritis It is well established that high doses of monomeric immunoglobulin G (IgG) purified from pooled human plasma [intravenous immunoglobulin (IVIG)] confer anti-inflammatory activity in a variety of autoimmune settings. However, exactly how those effects are mediated was not clear because of the heterogeneity of IVIG.
It is well established that high doses of monomeric immunoglobulin G (IgG) purified from pooled human plasma [intravenous immunoglobulin (IVIG)] confer anti-inflammatory activity in a variety of autoimmune settings. However, exactly how those effects are mediated was not clear because of the heterogeneity of IVIG.
Anti-inflammatory activity of IVIG/2,6-sialylated Fc is medicated by SIGNR1 and FcgRIIB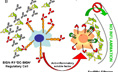 The anti-inflammatory activity of intravenous Ig (IVIG) results from a minor population of the pooled IgG molecules that contains terminal alpha2,6-sialic acid linkages on their Fc-linked glycans. These anti-inflammatory properties can be recapitulated with a fully recombinant preparation of appropriately sialylated IgG Fc fragments.
The anti-inflammatory activity of intravenous Ig (IVIG) results from a minor population of the pooled IgG molecules that contains terminal alpha2,6-sialic acid linkages on their Fc-linked glycans. These anti-inflammatory properties can be recapitulated with a fully recombinant preparation of appropriately sialylated IgG Fc fragments.
